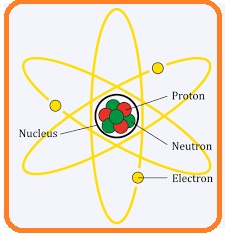
 Hello, friends welcome to the new post. in this post, we will have a detailed look at Difference Between Electron and Proton. The atom is the least portion of any substance that used to create an object. There are further three main sub-parts of the atoms first one is an electron second one is a proton and the third one is a neutron. Atoms is considered as the main element of any substance since it is created through the connection of different atoms. Atom has positive charge when its release electrons and has a negative charge when getting the electrons
Hello, friends welcome to the new post. in this post, we will have a detailed look at Difference Between Electron and Proton. The atom is the least portion of any substance that used to create an object. There are further three main sub-parts of the atoms first one is an electron second one is a proton and the third one is a neutron. Atoms is considered as the main element of any substance since it is created through the connection of different atoms. Atom has positive charge when its release electrons and has a negative charge when getting the electrons
There is a large number of atoms used to create molecules and these atoms are combine through chemical bonding. If we do compassion between electron and proton then come to know that there is positive charge at proton and negative at electrons. Both of these components are fundamental parts of atoms. So let’s get started.
Difference Between Electron and Proton
Introduction to Electrons
- The smallest part of atoms of any material that is denoted with the letter e and has a charge of negative value.
- It is part of a lepton group of different particles and it’s the main component of the structure of ant material.
- Its mass is 1/1836 time of protons mass that is also part of the atom and has a positive charge.
- It shows both behaviors like wave and particles when strike with other objects shows diffraction behavior like light.
- Its wave feature can be detected through different experiments.
- This article helps to sturdy the different educations lie electricity, chemistry, physics, etc.
- Due to the charge on the electron, there is an electric field exist about it and moving electron shows a magnetic field.

what is Protons
- The proton is also a particle of atoms that exists in the inner side of atoms in the nucleus and has a positive charge.
- Its mass is less than the neutron that exists with it in the nucleus.
- Singel or large protons exist in the nucleus The number of protons existing in the atoms tells about the atomic number that is denoted with the letter Z.
- Every element has a different value of Z due to different number of protons
- The atoms that have the same number of protons and neutrons that atoms has zero charge
- the ahrge at protons is 1.6021 x 10^-19
- Its removal and addition in the atoms is the difficult to process like the electron.
Electron and Proton
Electron
- The electron is sub atomic particle that comes with a negative charge.
- it is denoted with e letter.
- It has a negative polarity charge.
- Electron exists in space about the nucleus.
- It moves in a defined orbit about the nucleus of an atom
- It can be added or removed from an atom
- Electrons cause the flow of current
- The value of charge on an electron is -1.6 × 10-19 C.
- Its mass is 9.1 × 10-31 kg.
Proton
- The proton is also sub atomic particle that comes with postie charge.
- It is dentoed with p.
- its charge is +1.6 × 10-19 C.
- Its mass is 1.67 × 10-27 kg
- Proton exists in the nucleus atom
- Protons do not move since they are in the nucleus and bound with nuclear forces.
- It is difficult to add or remove protons.
What makes protons and electrons different?
- They come with charges protons have a positive charge and electrons have a negative charge.
What is a proton made of?
- Protons come with 2 up quarks and one down quark, and neutrons come with one up quark and 2 down quarks, These quarks are called valance.
Can protons and electrons be equal?
- The positive charge on a proton is equal to the magnitude of the negative charge on an electron. In a result, neutral atoms have an equal number of protons and electrons.
What is an example of a proton?
- The nucleus of a hydrogen atom or H+ ion is a proton. Each atom of hydrogen comes with one proton, every lithium has 2 protons, and lithium has 3 protons, etc.
Read also
that is a detailed post about the Difference Between an Electron and a Proton if you have any further queries ask in a comment, Thanks for reading. have a nice day.





