 Hi, friends welcome to the new post. Here we will learn the Difference Between Anode and a Cathode. Electrodes are the main element of any cell that is used to make a connection with other devices for the completion of circuits. There are two main categories of electrodes. The first one is the anode and the second one is the cathode.
Hi, friends welcome to the new post. Here we will learn the Difference Between Anode and a Cathode. Electrodes are the main element of any cell that is used to make a connection with other devices for the completion of circuits. There are two main categories of electrodes. The first one is the anode and the second one is the cathode.
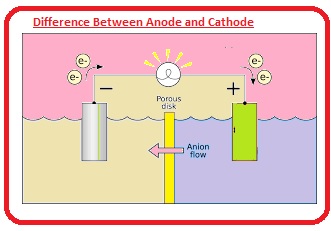
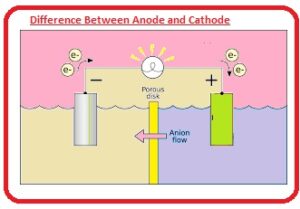
The anode is a positive terminal here oxidation process is done while the cathode is negative and reduction is done here. Here we will discuss the different parameters for these two modules in detail. So let’s get started.
Difference Between Anode and Cathode
What is Anode
- The category of anode through which current goes to the different electrical components that have a polar nature.
- The current passing in the diode has the nature of the conventional mean movement of positive charges.
- The oxidation process is done at the anode terminal example of this process is galvanic cells.
- The anode is also named as zincode.
- This terminal works as a donor since provides the electrons to the circuitry.
- In case of discharging of any module, the current is coming out from it.
- If a device is being charged then the current anode is working like the cathode.
- In primary types of cells, there are no polarity changes in the anode and cathode while in the case of secondary cells polarity reversal occurs.
What is Cathode
- It is a category of the electrode that has a negative polarity and conventional current that goes out from it.
- Conventional current is current that is produced due to the movement of anion or positive charges.
- Due to negative polarity electrons move in the reverse direction of conventional current.
- In the case of the galvanic cells shows positive behavior.
- Consumption of charges is done at his point.
- it shows attraction for positive charges and repulsion for negative ones.
- This terminal is also named an acceptor of electrons.
- The reduction process is done at this module.
- In chemistry subject the point of the cell where reduction exists.
- It is denoted with the letter C.
- Electrons are given to the positive charge ions that move to the electrolyte.
- The movement of electrons from the cathode to parts of the solution is called cathode current.
Anode and Cathode in Electrolysis
At the time of electrolysis chemical reactions or changes occur through the current flow in the circuit and the electrolytic cells are devices that transform electrical energy into chemical energy. The charge of anion and cation is positive and negative in electrolytic cells. let’s suppose electrolysis of molten NaCl.
The anode of the wire or plated has a positive charge since it is connected to the positive terminals of the battery. So anions move towards the anode and release electrons to the anode and get oxidized.
These electrons are provided by anion passing to the circuit. In this electrolysis of molten NaCl, 2 inert electrodes are placed in molten NaCl solution. If the current passes anode produces high positive charges and CL- ions from the solution move to the anode, where they oxidize to sodium metal.
Cathode connected with negative terminals of battery and have larger negative charges. Cations in solution move towards it and are reduced by getting electrons from the external circuit.
In a galvanic cell cation is a positive electrode if molten NaCl faces electrolysis when current flows. Na+ ions start moving to the cathode and are reduced to make sodium metal.
Anode vs Cathode
| Characteristic | Cathode | Anode |
| Definition | On the cathode gain of electrons or reduction process occurs | Here oxidation or loss of electrons occurs |
| Electron Flow | Electrons move into the cathode from an external circuit | It releases electrons |
Charge Accumulation | positive charge. | negative charge. |
Function in Cells | uses electrons for spontaneous electrochemical reactions. | electrons produce for spontaneous electrochemical reactions. |
Direction of Current Flow | cathode to anode. | anode to cathode. |
| Example | battery, where the cathode reduction occurs. | the anode in the battery where oxidation occurs during discharge. |
Faqs
Is a cathode positive or negative?
- The cathode is an electrode where a curent gives or flows externally. The anode is the positive side and the cathode is the negative side.
Which is called cathode?
- The cathode is a metallic electrode where current passes out in polarized electrical devices. While anode is an electrode in polarized electrical devices where current flows in from the external side.
How to identify anode and cathode?
- The positive electrode is an electrode having a high potential as compared to a negative electrode. At the time of discharge positive electrode is the cathode and the negative electrode is the anode.
- While the positive electrode is the anode and the negative electrode cathode.
Read also
That is a detailed post about the Difference Between an Anode and a Cathode. If you have any further queries ask in the comments. Thanks for reading have a good day.